Adenoviruses
Adenoviruses were first isolated from adenoids surgically removed from children in 1953 as part of a study to evaluate different tissues for growth of polio viruses by Rowe et al. They were soon established as the aetiological cause of acute respiratory disease. Adenoviruses have been isolated from every species of mammals, birds and amphibians studied.
A. Properties
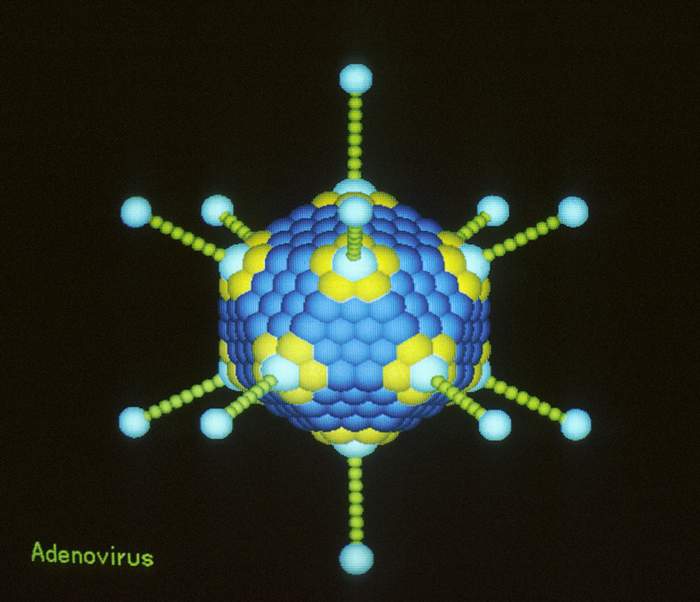
Naked double stranded DNA virus
Icosahedral symmetry, 80nm in diameter
Capsid consist of 252 capsomeres (240 hexons, 12 pentons)
Pentons make up the apices and possess projecting fibers
The fibers possess haemagglutinating activity and mediate the attachment of the virus to cellular receptors.
At least 47 serotypes of human adenoviruses are known and they are classified into 6 subgenera (A to F) on the basis of oncogenicity on newborn hamsters and nucleotide sequence homology
The hexons contain a family-reactive determinants, type-specific determinants are present on the pentons and hexons.
By 1989, 47 human adenoviruses had been identified. They were originally classified into subgenera on the basis of their oncogenicity on newborn hamsters. Subgenus A comprises of adenovirus serotypes that are highly oncogenic i.e. causing tumours in every animal within 2 months. Subgenus B adenoviruses are weakly oncogenic, meaning that few of the animals developed tumours after an observation period of one year. The nononcogenic adenoviruses can transform rodent cells in vitro and are divided into subgenera C and D on the basis of differences in the antigenicity of the early antigen. Classification was also made on the basis of haemagglutination of rat or Rhesus monkey erythrocytes. Further classification was made by SDS- PAGE of virion polypeptides and a further 2 subgenera were defined. (subgenus E - Ad4, subgenus F - Ad40, 41) In general though, the critical classification depends on nucleotide sequence homology between the genomes of different viruses. Studies showed that the degree of DNA homology between members of the same subgenus is above 50%, whereas the homology between members of different subgenera is below 20%.
In summary, members belonging to the genus adenoviruses share common epitopes on the hexons. Subgenus is defined by the DNA homology of more than 50% between members within a subgenus and less than 20% between members of different subgenera. The serotype is defined by quantitative neutralization with hyperimmune sera. The ratio of homologous to heterologous neutralization titre must be greater than 16.
Host Range - Human adenoviruses can infect a variety of laboratory animals, resulting in inapparent infections to tumours, depending on the type of adenovirus and the type of animal involved.
Cell Culture - A variety of mammalian cells support the multiplication of adenoviruses often producing characteristic cyptopathic effects. Rodent cells can be immortalized.
The recognized diseases of adenoviruses predominantly involve the respiratory tract, the GI tract and the eye. Virus may be introduced through contact, respiratory droplets or ingestion. The association of particular types with specific disease syndromes is striking. After recovery of illness, adenoviruses may maintain latent persistent infections in the tonsils, the adenoids, and other lymphoid tissues of man, and they are readily activated. (Most persons are infected with one or more types of adenovirus before the age of 15, 50 to 80% of tonsils removed surgically yield an adenovirus when cultured in vitro). Ad1, Ad2 and Ad5, members of subgenus C, persist in tonsils for several years. Shedding of infectious virus in the stools for at least 2 years have been documented. Adenovirus strains can also be secreted in the urine.
Adenoviruses are associated with a wide vareity of clinical syndromes, the majority of which concerns the respiratory tract.
| 1. | Pharyngitis | 1, 2, 3, 5, 7 |
| 2. | Pharyngoconjunctival fever | 3, 7 |
| 3. | Acute respiratory disease of recruits | 4, 7, 14, 21 |
| 4. | Pneumonia | 1, 2, 3, 7 |
| 5. | Follicular conjunctivitis | 3, 4, 11 |
| 6. | Epidemic keratoconjunctivitis | 8, 19, 37 |
| 7. | Petussis-like syndrome | 5 |
| 8. | Acute haemorrhaghic cystitis | 11, 21 |
| 9. | Acute infantile gastroenteritis | 40, 41 |
| 10. | Intussusception | 1, 2, 5 |
| 11. | Severe disease in AIDS and other immunocompromized patients | 5, 34, 35 |
| 12. | Meningitis | 3, 7 |
Respiratory Disease in Children - Adenoviruses are responsible for 5% of acute respiratory infections in children under the age of 4 years, whereas they account for 10% of hospitalized respiratory infections in this age group. Adenoviruses can also cause laryngotracheobronchitis, but the pneumonias that occur in young children are the most serious manifestations. These may occur as a consequence of infection with the endemic Ad2 and Ad5, among which certain strains can be more aggressive than others. In particular, Ad 3 and Ad7 may also create severe problems. Adenoviruses have been reported to account for 10% of pneumonias of childhood. The severity of symptoms is related to overcrowding. In the winter of 1959, 3398 cases of adenovirus pneumonia with a fatality rate of 15.5% were seen at the Peking children's hospital. Among the survivors of severe respiratory infections, residual lung damage due to secondary obliterative bronchiolitis has been reported. Bronchiectasis and abnormal lung function tests have also been reported as sequelae.
Pharyngoconjunctival Fever - This disease is characterized by conjunctivitis, fever, pharyngitis and adenoidal enlargements. This is frequently associated with swimming pools. Adequate levels of chlorine are usually sufficient to inhibit outbreaks.
Acute Respiratory Disease (ARD) in Military Recruits - ARD is usually caused by Ad4, Ad7, and Ad21, although Ad14 has been reported from Holland. In general, outbreaks do not involve seasoned troops but cause a high morbidity among newly enlisted troops. Adenovirus infections among healthy civilian adults are less common. The crowding of people, allowing repeated exposure to highly infectious doses, and the strenuous physical exercise may account for the unusually high degree of severe infections. ARD usually appears during the third week in training. Characteristic symptoms include fever, malaise, sore throat, hoarseness and cough. Pneumonia develops in around 10% of cases.
Pertussis - Adenoviruses can be isolated frequently (39%) of patients infected with B. Pertussis. The contribution, if any, of adenoviruses to the pertussis syndrome is not known.
Infections of the Eye - Acute follicular conjunctivitis, which is part of the syndrome of pharyngoconjunctival fever, can also occur as a separate entity. The disease is also associated with swimming pools. Epidemic keratoconjunctivitis is a distinctly different syndrome. This syndrome is characterized by an aggressive conjunctivitis, pain, photophobia and lymphadenopathy followed by the development of superficial punctate keratitis, corneal opacities may last for several years. In 1941, more than 10,000 cases occurred in the marine shipyards of Pearl Harbour ("Shipyard eye"). Adenovirus keratoconjunctivitis mainly affects males in Western countries or children in Asia and may last for 4 to 6 weeks. Antibody to Ad 8 is uncommon in American school children, whereas approximately half of the Japanese or Taiwanese children have such antibodies.
Acute Haemorrhagic Cystitis - This syndrome occurs predominantly in 6 to 15 year old boys. The syndrome consists of acute dysuria with haematuria and is mainly associated with Ad 11. Its significance lies in the potential of being confused with more serious diseases of the kidney such as glomerulonephritis.
Infections of the gut - Adenoviruses are associated with 4-15% of all children hospitalized with viral gastroenteritis. Gastroenteritis may be a sign of a systemic infection such as those caused by Ad3 or Ad7. They may cause both respiratory and diarrhoea in a child with high fever. However, the enteric adenoviruses Ad40 and Ad41 are associated with 2/3rds of cases of adenovirus-associated diarrhoea. Several reports have appeared implicating adenoviruses in mesenteric adenitis and intussusception of the gut. (For more information on enteric adenoviruses, please visit the webpage on diarrhoeal viruses)
Severe combined immunodeficiency (SCID) and immunocompromised hosts. - Children with SCID are prone to develop severe infections with the most frequently occurring persistent adenoviruses. Pneumonia and hepatitis, with a high mortality is seen.
AIDS - latent infections of the kidneys are occasionally seen in patients with AIDS and can be readily isolated from the urine. Members of subgenus D are often seen in the stools of AIDS patients.
Bone marrow transplant recipients - These
patients are vulnerable to activation of all
latent DNA viruses. Adenovirus infections have
been demonstrated in 8% of bone marrow transplant
recipients. There is evidence that adenovirus
infections are responsible for substantial
mortality but more work needs to be done in order
to assess the precise role of adenoviruses in
bone marrow transplant patients.
Virus Isolation - Adenovirus may be isolated from most bodily fluids and secretions; from eye swabs, NPA, Throat swabs, urine, faeces, leucocytes and the CSF. The most reliable source for isolation is faeces, particularly for members of subgenera A, D, and F. Members of subgenera B, C, and E can also be isolated form the urine. Adenoviruses are stable and do not need refrigeration during transport. HEK cells are permissive for growth of all adenovirus strains except the enteric strains Ad 40 and Ad41. Hep-2 cells can also be used. Primary monkey kidney cells and 293 cells will permit the growth and isolation of Ad 40 and Ad41 strains. Adenoviruses produce a characteristic CPE in cell culture, which consist of the rounding and clustering of cells with refractile intranuclear inclusion bodies. The CPE may start at the periphery of the culture before generalizing. However, some of the group B adenoviruses do not cause cell enlargement or clustering.
The isolate can be positively identified as an adenovirus by complement fluorescent antibody, complement fixation or ELISA tests, using antibodies against the common group antigen (EM may also be used). The adenovirus can be typed by the use of neutralization tests or HAI tests. (NB. The adenovirus CF antigen is a property of the hexon that contains group specific as well as type specific Ag. The HA antigen is carried by the pentons which has type-specific antigenic epitopes. Neutralization properties are intrinsic to both the external part of the hexon and the fiber) The FA test using a hyperimmune serum to any of the common adenovirus serotypes will react with the rest of the human adenoviruses. Recently, type-specific monoclonal antibodies have become available for use in fluorescent antibody, ELISA, and LA tests for differentiating enteric Ad 40 and Ad 41 from other adenovirus strains. The isolate can also be typed by hybridization and restriction endonuclease digestion patterns.
Detection of Ag by IF - Direct microscopic examination of exfoliated cells is not generally successful. However, with the use of IF techniques against the common adenovirus antigen, a sensitivity rate approaching that of routine tissue techniques have been reported. However, some researchers using IF have detected only one-third of the NPA from which adenoviruses were subsequently isolated in tissue culture. Furthermore, IF has not been useful in the early diagnosis of conjunctival adenovirus lesions but may be useful in diagnosing acute haemorrhagic cystitis.
PCR - PCR is becoming increasingly used for the rapid diagnosis of adenovirus infection.
Serology - Mammalian adenoviruses share group-specific antigen epitopes (on the inside of hexons) which is recognized by complement fixing antibodies. Complement fixation for adenovirus- specific antibodies is ideally performed with a pool of antigen representing a serotype of each subgenus. If this is hard to accomplish, representative serotypes of subgenus B and C should be used. The extract should be prepared from sonicated infected cells which will contain a 10 fold excess of free hexons. ELISAs have also been prepared against the common group antigen. Because many individuals would have experienced recent infection with another adenovirus, and thus already have a high titre of AB against the group Ag, it was suggested that CFTs will only detect 50% of new infections. To type the virus, HAI and neutralization tests would have to be used. For HAI tests, rat erythrocytes should be used for subgenus A, C, D, E or F, and monkey erythrocytes for subgenera B. Serum neutralization assays are cumbersome but are the most efficient means of detecting specific antibodies against each adenovirus serotype.
There is no specific antiviral chemotherapy against adenoviruses at present. Idoxuridine and ARA-A had been tried in the treatment of keratoconjunctivitis but were unsuccessful. Antivirals have not been tried for the adenovirus-induced respiratory syndromes. When these diseases occur, they are difficult to distinguish from similar diseases caused by a variety of RNA viruses. Swimming pool-associated conjunctivitis can be prevented with adequate levels of chlorine in the water.
Effective vaccines are available for use in the military against adult respiratory distress syndrome. These vaccines are not currently licensed for administration to civilians. Non- attenuated live Adenovirus types 4, 7, and occasionally 21 are packaged into enteric capsules so that they bypass the respiratory epithelium and only replicate once they reach the intestine, resulting in an asymptomatic infection of the intestine. These strains were cultivated in human embryo fibroblasts. Earlier adenovirus vaccines were grown in primary monkey cells that have been demonstrated to contain SV40 virus. SV40 virus genome can integrate into the E3 region of adenovirus, indeed a recombinant adenovirus was isolated which contained the tumourigenic T antigen of SV40. As a result, this approach to producing a vaccine was abandoned. At least 15 million doses of adenovirus vaccine had been given and the vaccine had been shown to be both safe and effective. Adenovirus-associated pneumonia has ceased to be a major problem in the US army. The vaccine has not been licensed for use in children because of worries over the lack of attenuation and the potential oncogenic potential of these vaccines.