 |
 |
Rabies
There are few viruses as successful as rabies virus. It has survived for millennia in most parts of the world. It can replicate in all warm-blooded animals, where the resultant disease is almost invariably fatal. The virus ensures its own survival by causing the afflicted host to find another host for it. It was thought that a single virus type is responsible for all the rabies diseases. It was not until the discovery of rabies-related viruses such as the Lagos bat, Mokola and Duvenhage viruses in the Lyassavirus genus, was this view seriously challenged.
A. Properties
member of the Lyassavirus of the Rhabdoviridae
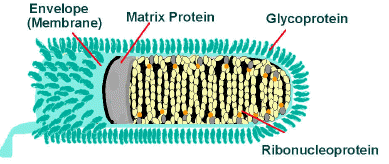
ssRNA enveloped virus, helical symmetry
infectivity destroyed by lipid solvents
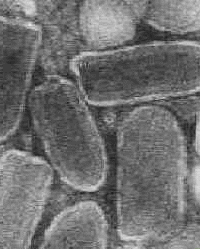
6-7 nm spike projections are present on the envelope
characteristic bullet-shaped appearance
virion 130-240nm * 80nm
-ve stranded RNA codes for 5 proteins; G, M, N, L, S
Exceeding wide range of hosts
Electronmicrograph and schematic diagram of rabies virus particles (Source: CDC)
Rabies virus have been adapted to growth in a wide variety of primary and continuous cell systems, not only from cells of warm blooded animals but also ones of poikilothermic vertebrate origin. The virus is grown in human diploid cells for the purpose of producing a vaccine. It has also been adapted to growth in avian embryos. Monoclonal and polyclonal studies of rabies isolates from many animal studies worldwide have led to the following classification of the rabies group of Rhabdoviridae, genus Lyssavirus. Lagos bat, Mokola and Duvenhage viruses have been isolated from various animals in a number of African countries. Their natural hosts are unknown and are thought to be bats or rodents. EBL-1 and EBL-2 have been isolated from European bats. Duvenhage and EBL-2 viruses have been associated with human infection that resulted in a rabies-like illness and death.
| Serotype 1 | prototype rabies virus |
| Serotype 2 |
|
| Serotype 3 | Mokola |
| Serotype 4 | Duvenhage |
| Serotype 5 | EBL-1 ((European bat lyssaviruses) |
| Serotype 6 | EBL-2 |
Rabies is a zoonosis which is prevalent in wildlife. The main animals involved differs from continent to continent.
| Europe | fox, bats |
| Middle East | wolf, dog |
| Asia | dog |
| Africa | dog, mongoose, antelope |
| N America | foxes, skunks, raccoons, insectivorous bats |
| S America | dog, vampire bats |
Increasingly, bats have been recognized as important reservoirs of rabies in many parts of the world. Cases of rabies in humans have been reported after bites by bats. Rabies virus had been shown to infect all mammals so far tested. Dogs, cats and cattle are particularly susceptible. Skunks, bats, foxes, squirrels, badgers, raccoons and mongooses are the principle wildlife host. Foxes are the main carriers in Europe. Birds have also been shown to be susceptible to infection. Compartmentation occurs with rabies, so that the disease is reported in one major host species in certain geographical areas while it is reported less frequently in the same species in other areas of endemic rabies.
The commonest mode of transmission in man is by the bite of a
rabid animal or the contamination of scratch wounds by virus-
infected saliva. However, other routes have been implicated in
the past, such as through mucous membranes of the mouth,
conjunctiva, anus and genitalia. Infection by aerosol
transmission had been demonstrated in experimental animals and
has been implicated in human infection in rabies-infected bat
caverns and in several laboratory accidents. Man to man
transmission by transplantation of infected corneas were reported
in 5 instances. Rabies is an acute infection of the CNS which is
almost invariably fatal. The virus is similar to VSV of cattle.
Following inoculation, the virus replicates in the striated or
connective tissue at the site of inoculation and enters the
peripheral nerves through the neuromuscular junction. It then
spreads to the CNS in the endoneurium of the Schwann cells.
Terminally, there is widespread CNS involvement but few neurons
infected with the virus show structural abnormalities. The nature
of the profound disorder is still not understood.
The incubation period is highly variable, ranging from 7 days to several years. It depends on several factors such as;
1. Dose of inoculum
2. The severity of the wound
3. The length of the neural path from the wound to the brain e.g. wounds on the face have a shorter incubation period than wounds in the leg.
The illness begins with a non-specific prodrome period, comprising of fever, malaise, anorexia, N+V, sore throat, myalgia and headache. The patient nay exhibit irritability and abnormal sensations around the wound. The prodrome is followed by one of two basic clinical patterns: the more common "furious" form characterized by hyperexcitability, spasms and hydrophobia; or "dumb" rabies featuring an ascending paralysis. Survival tends to be longer for patients with "dumb" rabies than those with "furious" rabies.
Complications involving the Cardiovacular System, CNS, and the
respiratory systems eventually develop and contribute to death.
Cardiac dysrhythmias of all types occur and respiratory
disturbances occur in all cases. Raised intracranial pressure
contributes to the decreased level of consciousness and to focal
convulsions. Other CNS complications include disturbances of
thermoregulation, diabetes insipidus, autonomic dysfunction and
convulsions. The differential diagnosis of rabies includes
tetanus, poliomyelitis, Guillain-Barre syndrome, viral
encephalitis and poisonings and drugs.
The diagnosis of animal and human rabies can be made by 4 methods: (1) histopathology (2) virus cultivation (3) Serology (4) virus antigen detection. Although each of the first 3 methods have distinct advantages, none provide a rapid definitive diagnosis.
Histopathology - Negri bodies are pathognomonic of
rabies. However, Negri bodies are only present in 71% of
cases.
Virus cultivation - The most definitive means of
diagnosis is by virus cultivation from infected tissue.
Tissue culture lines, such as WI-38, BHK-21, or CER.
Since rabiesvirus induce minimal CPE, IF is routinely
used to detect the presence of rabiesvirus Ag in the
tissue culture. The more commonly used method for virus
isolation is by the inoculation of saliva, salivary gland
tissue and brain tissue intracerebrally into infant mice.
The mice should develop paralysis and death within 28
days. Upon death, the brains are examined for the
presence of the virus by immunofluorescence.
Serology - circulating antibodies appear slowly in
the course of infection but they are usually present by
the time of onset of clinical symptoms. The most commonly
used serological tests were the mouse infection
neutralization test (MNT) or the rapid fluorescent focus
inhibition test (RFFIT). These tests have now been
largely superseded by EIAs. Serology had been reported to
be the most useful method for the diagnosis of rabies.
Rapid virus antigen detection - in recent years,
virus antigen detection by IF had become widely used. The
potentially infected tissue is incubated with
fluorescein-labeled antibody. The cells are examined by
fluorescent microscopy for the presence of fluorescent
intracytoplasmic inclusions. The specimens which are
usually used are corneal impressions (obtained by gently
abrading the cornea with a microscopic slide) or neck
skin biopsy (the cells examined are the sensory nerves).
In an American series, IF of corneal impressions or neck
skin impressions was diagnostic only in 50% of cases
early in the course of the clinical illness.
RT-PCR - this is now the test of choice for the
rapid diagnosis of rabies infection. It can be used on skin and saliva
samples.
Negri body in body of neuron and positive IF test for rabies antigen (Source: CDC)
Once rabies is established, there is nothing much that could be done except intensive supportive care. To date, only 2 persons with proven rabies have survived, and 3 persons with probable rabies. However, one survivor was left with severe neurological sequelae and all 3 who recovered were vaccinated beforehand. Numerous antiviral agents have been tried with no success.
1. Postexposure prophylaxis
In cases of animal bites, dogs and cats in a rabies endemic area should beheld for 10 days for observation. If signs develop, they should be killed and their tissue examined in the laboratory. Wild animals are not observed but if captured, the animal should be killed and examined. The essential components of postexposure prophylaxis are the local treatment of wounds and active and passive immunization.
Wound treatment - surgical debridement should be carried out. The wound should not be sutured up. Experimentally, the incidence of rabies in animals can be reduced by local treatment alone.
Passive immunization - human rabies immunoglobulin around the area of the wound; to be supplemented with an i.m. dose to confer short term protection. There is convincing evidence that combined treatment with rabies immunoglobulin and active immunization is much more effective than active immunization alone. Equine rabies immunoglobulin (ERIG) is available in many countries and is considerably cheaper than HRIG.
Active immunization - the human diploid cell vaccine is the best preparation available. The vaccine is usually administered into the deltoid region, and 5 doses are usually given.
2. Preexposure prophylaxis
Persons who are regularly at high risk of exposure, such as vets, laboratory workers, animal handlers and wildlife officers should be considered for preexposure prophylaxis by active immunization with the cell culture vaccine. Immunization normally consists of 3 doses of vaccine. Antibody can be demonstrated in the sera of virtually 100% of those vaccinated if the diploid cell culture vaccine is used. Booster doses should be offered to persons at continuing risk every one to three years. Local treatment of wounds should always be carried out in exposed persons who have been vaccinated previously. The WHO expert committee considers that local infiltration with antiserum is optional and systemic passive immunization contraindicated.
3. Rabies Vaccines
Several types of live attenuated vaccines are available for use in animals, but they are considered to be unsuitable for humans. The vaccines which are available for humans are present are inactivated whole virus vaccines.
Nervous tissue preparation - this consisted of a
5% suspension of infected animal nervous tissue which had
been inactivated (eg. the Semple vaccine was derived from
phenol-inactivated infected rabbit brain), These
preparations are now out of date as they were associated
with the rare complication of demyelinating allergic
encephalitis. This appears to be related to myelin basic
protein in the vaccine. This complication was shown to
occur in 4.6 case for 1000 persons vaccinated by the
Semple vaccine. The case-fatality proportion was 3.13%.
The Semple vaccine is still used in some developing
countries. A suckling mouse brain vaccine is used in some
Central and S.American countries.
Duck Embryo Vaccine - this vaccine strain is grown
in embryonated duck eggs and is inactivated with
B-propriolactone. This vaccine has a lower risk of
allergic encephalitis. However, it is considerably less
immunogenic and does have minor side effects. Almost all
vaccinees experience local reactions, 33% have
constitutional symptoms such as fever, malaise, myalgia,
and generalized lymphadenopathy.
Human Diploid Cell Vaccine (HDCV) - HDCV was introduced in 1978. It is a grown on WI-38 (U.S.) or MRC-5 (Europe) cells. The vaccine is highly effective, in several studies, antibodies have been demonstrated in 100% of all recipients. Serious adverse reactions to HDCV are extremely rare. However, the vaccine is very expensive ( $100 for 6 doses), as human cell cultures are more difficult to handle than other animal cell culture systems. 5 or 6 doses of the vaccine is normally i.m. However, several studies suggest than smaller intradermal doses of HDCV may be as effective and thus it may be considered for use in poor developing countries.
Another inactivated vaccine is widely used in China. It is derived from virus grown in primary hamster kidney cells and cost less than the other diploid cell culture vaccines. The vaccine had been shown to be as effective as HDCV. Efforts are being made to use other inexpensive cell culture systems such as VERO cells.
4. Failure of prophylaxis
HDCV has been used to treat many thousands of people exposed to possible rabid animals in the past 12 years and its efficacy has been proven. At least 16 people treated with HDCV after exposure have died of rabies. All of the patients had major exposures and in the majority, the incubation period was short, 21 days or less. Treatment was frequently not started promptly within 24 hours and only half received combined serum and vaccine. At least one person has died despite optimum treatment.
Urban - canine rabies accounts for more than 99% of all human rabies cases and over 90% of all human post-exposure treatments worldwide. In the past, the Scandinavian countries were able to rid themselves of rabies by sanitary control alone, which included stray dog control. Other countries, such as the UK, have used these techniques allied with quarantine and/or vaccination to eradicate and then maintain freedom from the disease. Currently, the importation of mammals into the UK is controlled by the Rabies order. It applies to a wide range of mammals but livestock including horses, which are covered by separate regulations. The animals must be vaccinated on arrival. Effective animal vaccines are available.
Wildlife - canine rabies can be controlled because in general, dogs live in close association with man and are therefore within physical reach. An attempt was made to vaccinate foxes in an attempt to create an immune barrier at the entrance to the Rhone valley in 1978. The live attenuated virus was contained in small plastic blister packages fixed under the skin of chicken heads used as a bait, and 4050 of these were distributed over an area of 335km2. With continued field trials, Switzerland has been freed of rabies. Other field trials are being set up.